
Holland & Barrett - Jacob Hooy CBD Olie! Nu 2e Halve Prijs. Shop in de winkel of online: http://bit.ly/2hhc50F #cbdolie #cbd | Facebook

AH Goudse Jong T-m Belegen Plakken En Stukken Van De Zelfbedieningsafdeling: 2e Halve Prijs Aanbieding bij Albert Heijn - AanbiedingenFolders.nl

Premium bonuskaart. AH Pancakes 2e halve prijs, Goudse Graskaas 1+1 gratis, Geraspte kaas 20%, AH 100% pindkaas 15%, Alpro Mild 20%, AH Gatenkaas 20%, Vivera kip 20%, Schotse Zalm 0.90, Iglo Ocean

Use the following half-reactions and the data in Table 19.1 to write the spontaneous reaction. Write the equilibrium law for this reaction and use the standard cell voltage to determine the value

Jumbo Amsterdamseweg Arnhem - EXTRA GOEDKOOP! Coca-Cola, Fanta, Sprite of Fernandes, nu de 2e verpakking voor de halve prijs! | Facebook
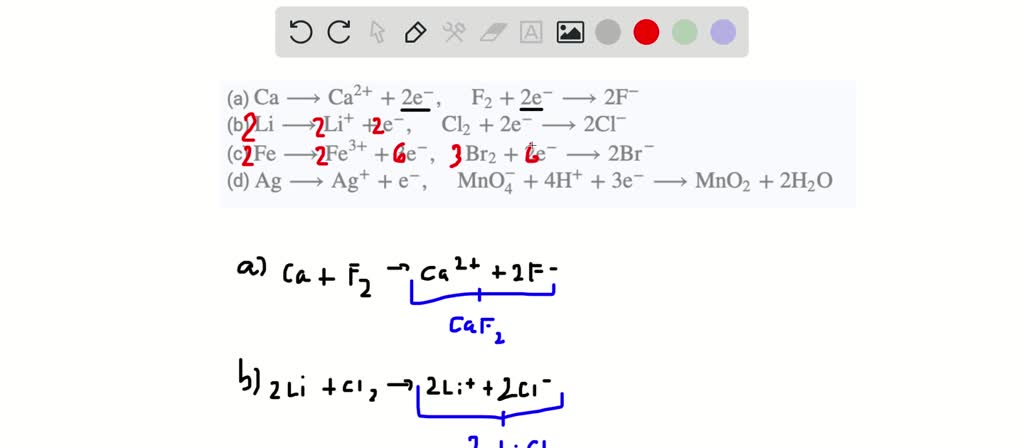
SOLVED:Assuming each pair of half-reactions below takes place in an acidic solution, write a balanced equation for the overall reaction. (a) Ca ⟶Ca^2++2 e^-, F2+2 e^- ⟶2 F^- (b) Li ⟶Li^++e^-, Cl2+2












![Sour Diesel [500mg] (sativa) vape - STIIIZY Sour Diesel [500mg] (sativa) vape - STIIIZY](https://product-assets.iheartjane.com/photos/89/2e/892ee2fb-9a20-44e9-bcbb-003da36bd02f.jpeg)



