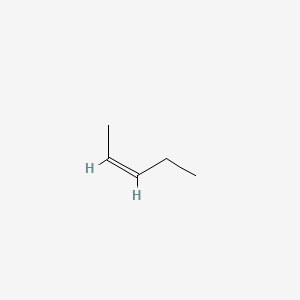
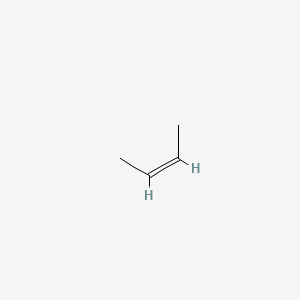
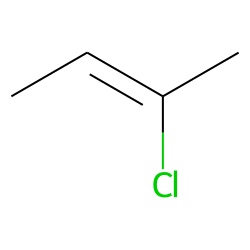
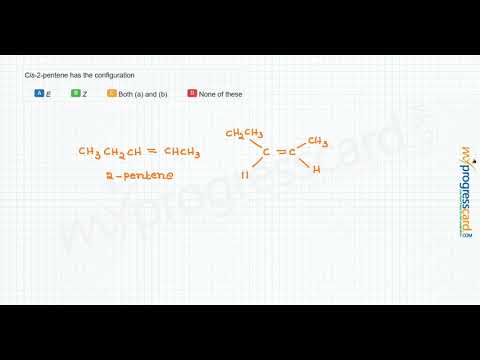
A)Draw the mechanism of bromination of cis-2-butene. B)Explain the stepwise mechanism of the reaction between mercury (II) acetate to ethylene C)Explain with suitable examples the concept of E-and Z- | Homework.Study.com

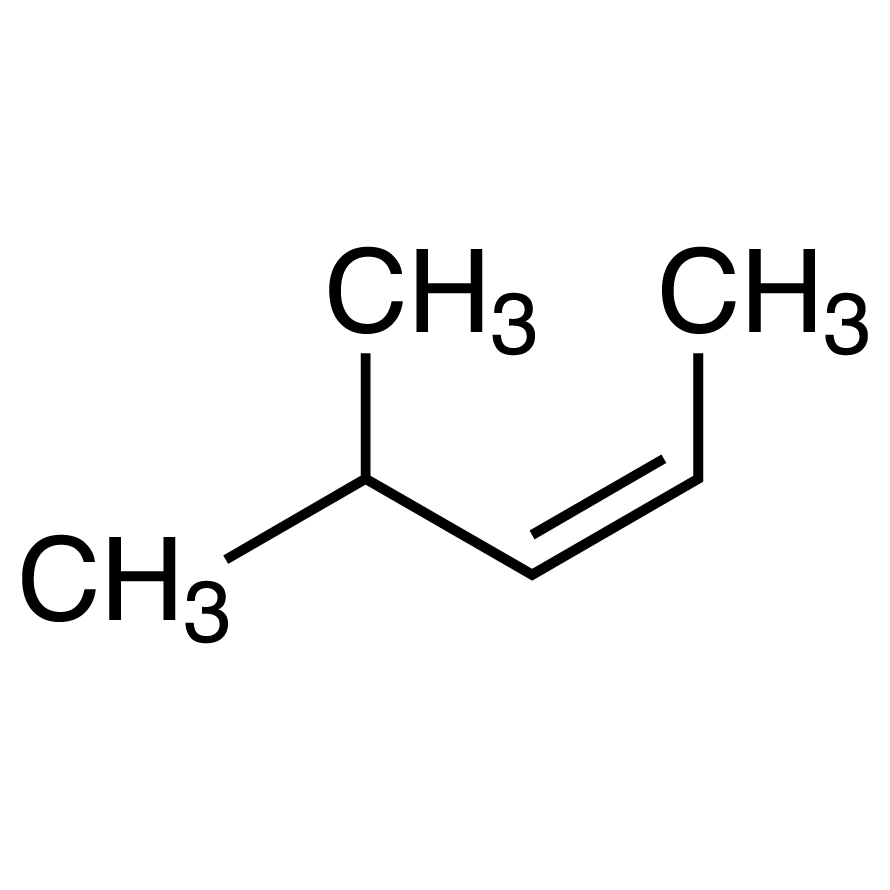
4-Methyl-2-pentene (cis- and trans- mixture) 96.0 %, TCI America, Quantity: 1 mL | Fisher Scientific